The reason why 'dendritic crystals' (dendrites), which hinder the practical application of the dream all-solid-state battery, are formed has been revealed.

Conventional lithium-ion batteries use a liquid electrolyte to fill the space between
Mechanically driven Li dendrite penetration in garnet solid electrolyte | Nature
https://www.nature.com/articles/s41586-026-10415-9
Understanding the short circuit in solid-state batteries
https://www.mpie.de/5151287/short-circuit-solid-state-batteries
Today, a wide range of products, from smartphones to electric vehicles, rely on batteries, and the energy storage capacity, lifespan, and safety of batteries will greatly influence the future of electrification. Among the next-generation battery technologies, one that is considered promising for practical application is the all-solid-state battery, which replaces the normally liquid electrolyte with a solid one.
Solid-state batteries use a solid electrolyte, enabling improvements in energy density, safety, and extended battery life. It is expected that once solid-state batteries become practical, smartphones could be used for several days without daily charging, and the driving range of electric vehicles could be tripled.
However, all-solid-state batteries have a problem: 'During charging, tiny needle-like structures called dendrites grow from the anode, penetrate the solid electrolyte, and connect to the cathode, causing a short circuit within the battery.'
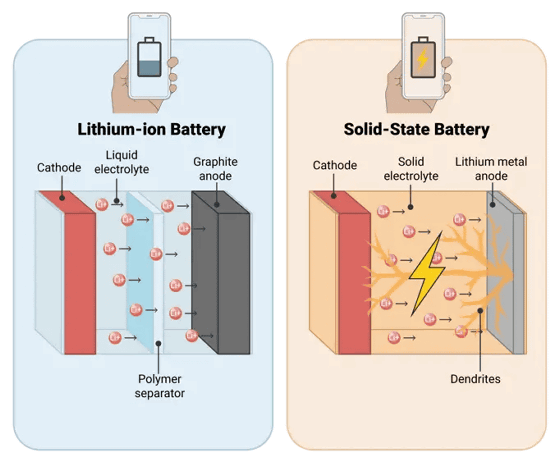
The following diagram shows a conventional lithium-ion battery using a liquid electrolyte on the left and an all-solid-state battery on the right. It shows that in the all-solid-state battery, dendrites grow from the anode and come into contact with the cathode, causing a short circuit.
The formation of dendrites in all-solid-state batteries is a counterintuitive phenomenon. Yuwei Zhang, a researcher at the Max Planck Institute for Sustainable Materials and lead author of the paper, states, 'The electrodes and the dendrites that are formed are made of lithium metal that is as soft as gummy candy, yet the dendrites can penetrate the ceramic electrolyte and cause a short circuit. How can soft dendrites break through hard solid ceramics?'
There are two theories regarding the mechanism by which soft dendrites penetrate hard solid electrolytes: one is that 'electrons leak along the grain boundaries (interfaces between crystals) of the solid electrolyte, promoting the formation of lithium nuclei that later interconnect,' and the other is that 'stress accumulates inside the dendrites, causing mechanical failure of the solid electrolyte.'
To test these hypotheses, the research team used complex sample preparation and characterization techniques to characterize the samples under vacuum and cryogenic conditions, unaffected by oxygen, water, or electron beams. As a result, no lithium concentration was observed beyond the dendrite tips, refuting the theory that electrons leak along grain boundaries.
On the other hand, analysis of the dendrites trapped within the cracks revealed that the precipitated lithium generated considerable hydrostatic stress , which in turn caused tensile stress in the solid electrolyte. This supported the theory that the stress inside the dendrites mechanically destroyed the solid electrolyte.
Mr. Chang stated, 'Soft lithium can penetrate hard ceramic electrolytes like a continuous water jet penetrating rock. Our calculations show that hydrostatic stress inside the dendrites ultimately causes brittle fracture of the solid electrolyte.'
The research team, which has elucidated the mechanism by which dendrites cause short circuits, is now exploring ways to prevent them. Possible approaches include 'increasing the toughness of the solid electrolyte to delay crack formation,' 'creating microscopic gaps to change the direction of dendrite growth,' and 'coating the electrodes to suppress dendrite growth.'
Related Posts:
in Science, Posted by log1h_ik